
Streamlining clinical workflows
and
patient data integration
The SparkCare® healthcare informatics integration tool simplifies patient assessment management and optimizes clinical workflows by integrating patient data collection technologies. It enables continuous, efficient data gathering during clinical research and practice, reducing administrative tasks and allowing more focus on patient care.
Clinicians and researchers can rely on SparkCare to securely and efficiently monitor patient assessment results. With the ability to create and customize various assessments for patients to complete remotely on their own devices or through NIH Toolbox®, SparkCare ensures comprehensive data collection. Once assessments are completed, results—including visuals, trends, and resources—are immediately available for clinician review and evaluation.
Integrating SparkCare within existing clinical workflows minimizes manual data entry, reducing errors and improving patient care while saving time and money. Patient health information and medical history can be continuously monitored and recorded over extended periods. The SparkCare healthcare informatics integration tool also accelerates the incorporation of newly acquired data into existing electronic health records (EHRs).
SparkCare was originally created through collaboration with the National Cancer Institute to help monitor and understand cognitive changes related to cancer and its treatment. SparkCare has evolved to now focus research and development goals on enhancing the availability and quality of all forms of clinical health data. We achieve this by integrating and harmonizing data across various tools and platforms to provide deeper insights.
SparkCare is a comprehensive data hub, connecting clinics with advanced tools to improve patient care and health outcomes.
Key features
Three easy-to-use, integrated components for clinicians and patients
Customized cognitive and behavioral assessments
Clinicians can create and customize assessments for patients to complete remotely on their own devices or through NIH Toolbox®.
Automated assessment ordering
A clinician portal that streamlines ordering assessments and automatically sends notifications to patients to complete them.
Integrates with mobile health-tracking apps and wearables
Secure, automatic streaming of PROMIS® and health data (Google Fit and Apple Health), as well as data from smartwatches.
HITRUST (Health Information Trust Alliance) compliance
System designed for HITRUST and HIPAA compliance.
Longitudinal report generation
Provides a rapidly readable report depicting patient data and trends, with customizable protocol-based recommendations for follow-up assessment.
Existing workflow integration
Supports in-clinic or at-home assessment measurements on various devices and integrates with existing EHRs.
Raw data access and download
Clinicians and researchers can securely access raw data.


SparkCare integrates with existing workflows, including NIH Toolbox, for seamless assessment administration and results integration.
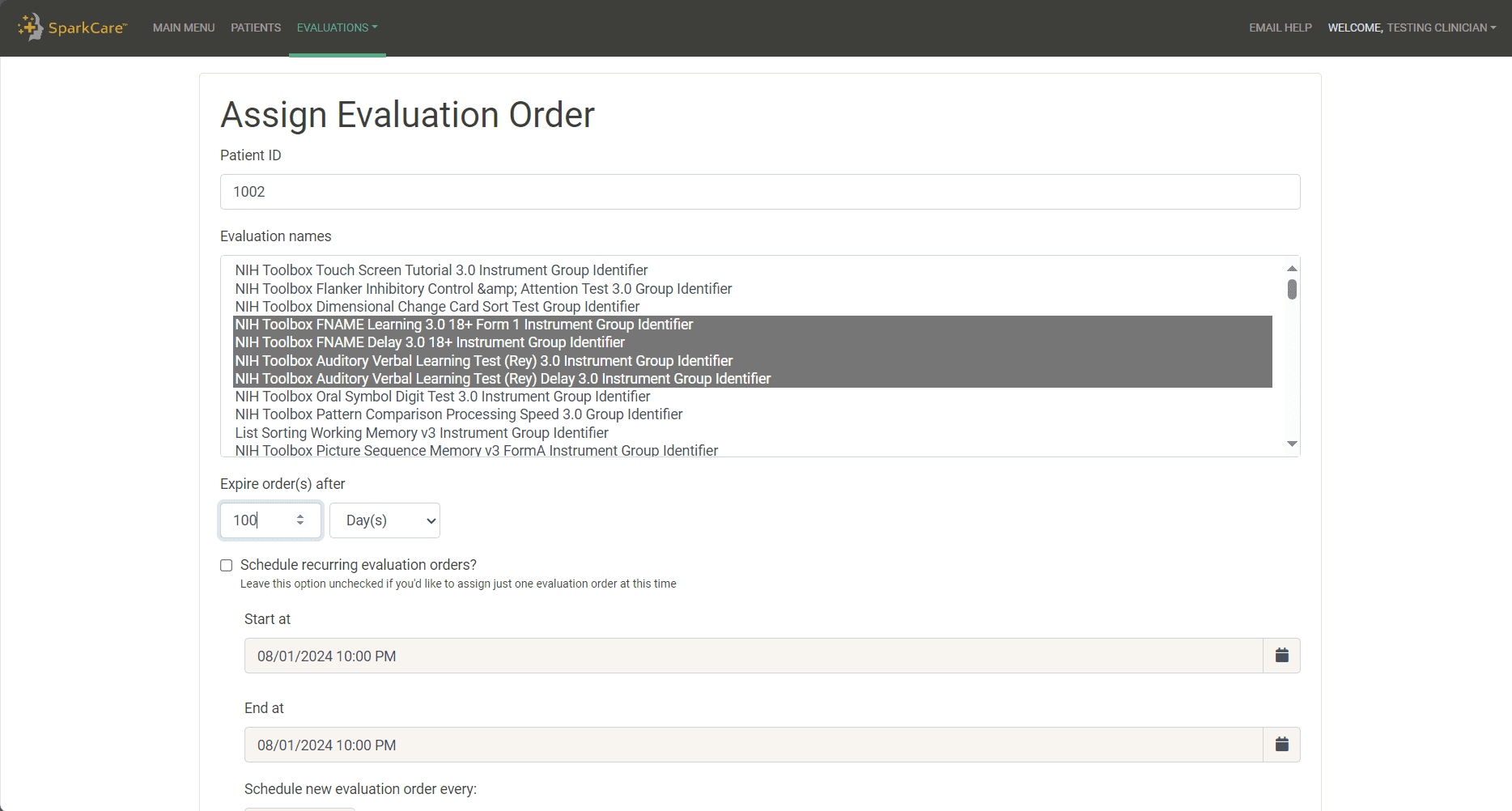
Create a new evaluation order or edit an existing one. Evaluation orders are bundles of assessments that are sent to the patient.
Send an evaluation order to the patient via email, so the patient can complete the assessment remotely.
The Platform
The SparkCare healthcare informatic integration platform is built on top of an extensible architecture that allows new interfaces to be brought online as needed. In addition to cognitive and behavioral assessments, Charles River Analytics is pursuing extensions to the SparkCare portal to include assessments for mild traumatic brain injury (mTBI), musculoskeletal injuries, and rehabilitation exercises. In addition, we hope to integrate our solution that enables patients to manage and adhere to clinician healthcare treatment plans.
Along with the ability to align with standard healthcare protocols and interface with existing EHRs, SparkCare aims to enable clinicians and researchers to obtain data from patients while outside the clinic, summarize the data in intuitive graphs and charts, and link the results to resources for providing follow-up care. Ultimately, SparkCare will extend the reach of the clinic to at-home assessments and tools to help manage and monitor changes in health status over time.

SparkCare provides cross-device support across users and contexts. (Desktop) Clinician-facing desktop portal to order assessments and review detailed results. (Mobile) Information exchange with NIH Toolbox for related assessment administration and results upload on tablet. (Tablet) Assessment link enables patients to take assessments on mobile device or any device with a web browser.
System requirements
The requirements necessary to successfully install SparkCare on a Windows Server are as follows.
- Windows Server (x86-64)
- PostgreSQL 12 or later
- pgAdmin4 6 or later
- Web browser
- Tablet/Mobile for administering the NIH Toolbox®
- NIH Toolbox and/or PROMIS® or license, obtained from HealthMeasures
Discover how SparkCare® can transform your clinical workflows and enhance patient care. Contact us today for a demo!


