
A system to practice resisting your cigarette cravings in a virtual world
A system to practice resisting your cigarette cravings in a virtual world
Constructed Environments for Successfully Sustaining Abstinence Through Immersive and On-Demand Treatment (CESSATION)
CESSATION is a narrative virtual reality (VR) tool that guides the user through a series of daily tasks while presenting cues that stimulate cigarette cravings. Users receive smoking-related cues (e.g., walking by a group of smokers) and experience common triggering events (e.g., their first cup of coffee, stressful work situations) so they can practice techniques to resist cravings in an immersive environment that feels much like the real world.
CESSATION is a TheraVerse® product.

“Current methods for smoking cessation are effective, but the odds of relapse remain high. Our CESSATION app helps smokers practice skills to avoid relapse within VR. The goal of CESSATION is to maximize efficacy of existing behavioral therapies, making them more immersive and accessible, both to widen the audience and to encourage more frequent and consistent use.”

Dr. Bethany Bracken
Principal Scientist and Principal Investigator for CESSATION
During standard treatment, a smoker practices resisting cravings in a group session, or by handling cigarettes and smoking paraphernalia in a clinician’s office. But when a smoker is faced with temptations in the real world (like when a co-worker offers a cigarette after a stressful shift at work), all that practice can drop away. The environment and cues vary dramatically between an artificial experience in an office, and the real world, making it challenging to enact the behaviors learned at the clinic in real life.
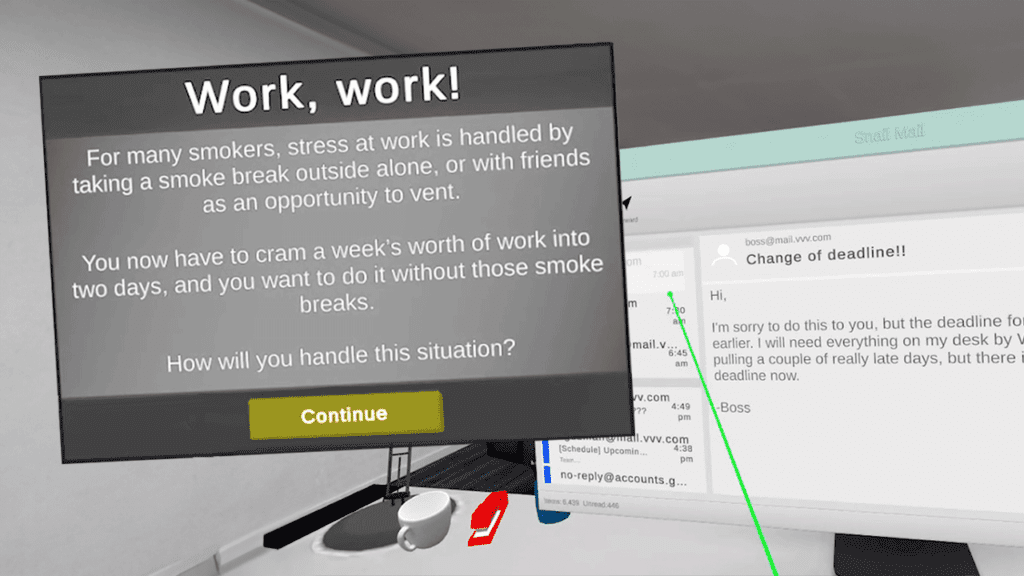



CESSATION presents its users with smoking-related stimuli in an immersive VR environment, allowing them to practice resisting cravings.
CESSATION, a TheraVerse® product, was developed with proven narrative game design techniques to produce a single, focused experience that holds a smoker’s interest even when used multiple times a day. Gaming and motivational psychology principles include trivia games and trophies.
CESSATION was tested on volunteer smokers in coordination with MedStar, who routinely conducts studies required for FDA clearance of new health systems. Each user attempted four narrative phases: at home in the morning, in a parking lot, in the office, and at home after work. At each phase, users were prompted to complete a series of everyday tasks while being presented with standard cognitive behavioral therapy techniques (designed with help from clinician partners Dr. Eden Evins and Dr. Corinne Cather at Massachusetts General Hospital) to resist cravings and handle stressful events without smoking.
“The amount of positive feedback we received positions CESSATION well for the next step, which is additional narrative and clinical content, and a full clinical trial,” said Dr. Bethany Bracken, Principal Scientist at Charles River and Principal Investigator for the CESSATION effort. “The study with MedStar allowed us to make important enhancements to our system and narratives. For example, participants were very clear about naïve VR users needing simpler controls, and some people reported difficulty standing for long periods of time. These results will guide future development of VR systems extending our narrative and therapeutic content to help people with all sorts of issues, from anger management to eating disorders.”
Charles River’s main goal for CESSATION is to design a tool that is accessible to as many people as possible. One of the benefits of VR treatment is that patients can use the technology directly without having to wait for their next appointment or group meeting. CESSATION removes the cost and time barriers of regular medical appointments, requiring only the patient and their own motivation to succeed.
CESSATION has the potential to help millions of people across the US who are trying to overcome substance dependence. Although the current release targets smokers, future adaptions of the core narrative framework and cues are currently being applied to vaped nicotine, with a plan to extend to alcohol and drug use, as well as to therapy for psychological disorders, like PTSD.
The CESSATION app can be downloaded from the Meta Quest Store.
Contact us to learn more about CESSATION, TheraVerse, and our other health and medical capabilities.
This project has been funded in whole or in part with Federal funds from the National Institute on Drug Abuse, National Institutes of Health, Department of Health and Human Services, under Contract No. HHSN271201700037C. Any opinions, findings and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Institute on Drug Abuse, National Institutes of Health, or Department of Health and Human Services.



